More well thought out work can be found at — https://axial.substack.com/
Axial partners with great founders and inventors. We invest in early-stage life sciences companies often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
Observations #32
A set of ideas and observations from a week’s worth of work analyzing businesses and technologies.
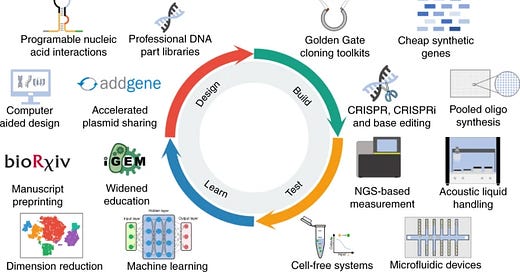
The second decade of synthetic biology
A review by Tom Ellis (UCL) does a great job summarizing progress in synthetic biology over the last decade - https://www.nature.com/articles/s41467-020-19092-2 Some key points:
A key paper came out in 2016 around genetic circuit design - https://science.sciencemag.org/content/352/6281/aac7341 The work came out of the Voigt Lab (MIT) with the lead author being Alec Neilsen (Asimov). The paper invented an end-to-end system called Cello to design circuits in E. coli that are insulated with minimal cross-talk. This paper was a powerful case study on the power of synthetic biology. It’s on par with the repressilator paper in the 2000s.
TALENs (TAL-Effector Nucleases) had a major impact on synthetic biology by enabling people to edit specific genes and binding specific regions of the genome. Cloning TALENS is really hard; I spent over a year cloning one in ~2012. Then CRISPR gene editing was discovered a few years later and got rid of the cloning process. But TALENS first showed the power of gene editing in synthetic biology.
Synthesis costs dropped driven by improvements in printing arrays and the decreasing costs of sequencing, which made QC of the DNA arrays much easier
Decreasing synthesis and sequencing allowed more labs and companies to finally use the DBTL framework in a cost effective way
De novo design of proteins saw a lot of progress mainly driven by the Baker Lab at UoW. The next decade will see more applications come out of this work.
Finally, healthcare became a larger focus for synthetic biology. Whereas metabolic engineering and using biology to make chemicals was the major focus in the 2000s.
Progress in science
With that overview in synthetic biology, a key lesson is that improved tooling enables new applications. A big shift over the last decade in synthetic biology was realizing its potential in healthcare beyond metabolic engineering. Fittingly, a quote from Syndney Brenner sums this up in one sentence:
“Progress in science depends on new techniques, new discoveries, and new ideas, probably in that order.”
- Sydney Brenner
ASO medicines
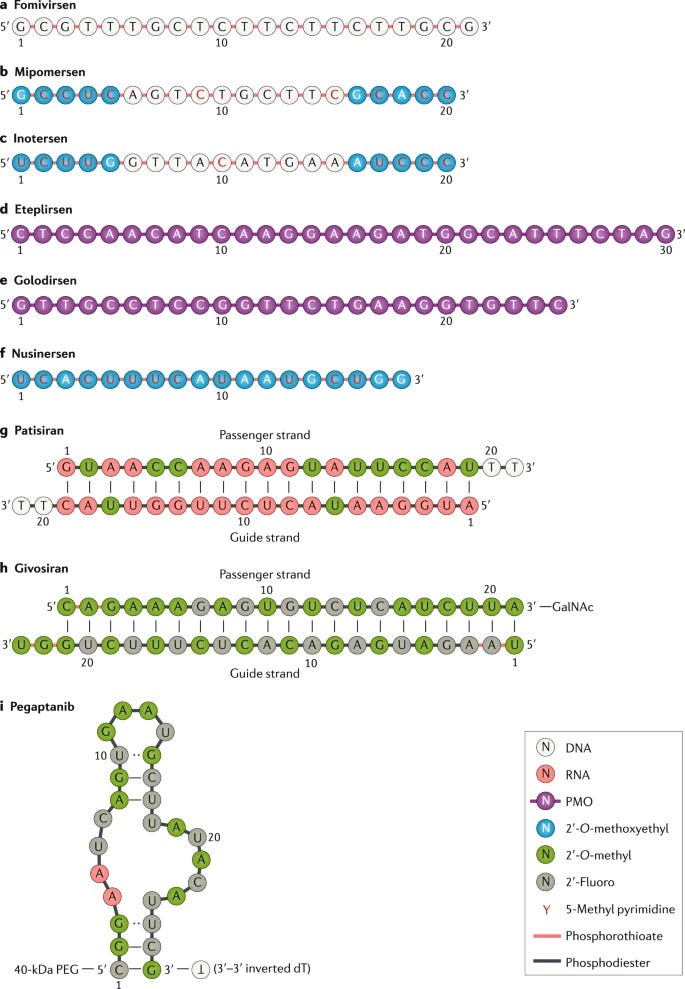
Antisense oligonucleotides (ASO) are synthetic single-stranded nucleic acids around 18 to 30 nucleotides to control gene expression of specific targets. Gene expression can be controlled through complete silencing, splice modulation, and even activation. ASOs interact with their corresponding targets through complementary Watson–Crick base pairing. This mechanism for ASOs is pretty powerful: leads are generated pretty easily once the sequence of the target is identified. With sequencing costs so low, the cost to generate an ASO lead is approaching 0. With this advantage, many ASO leads can be screened and optimized with medchem. However, a major bottleneck for ASOs has been selective delivery. ASOs have been really useful to cure liver disease because everything in the body gets filtered by the liver. The key strategy to solve the delivery problem is increasing the drug-likeness of an ASO:
Chemical modification of the ASO backbone to improve their ability to enter a cell and remain stable
Conjugation of an ASO to a cell-targeting modality (i.e. Avidity Biosciences)
Nanoparticles and exosomes as delivery vehicles. LNPs and viruses have also been used.
Despite this limitation, ASOs are an increasingly powerful modality due to:
Recent clinical and commercial success (i.e. Spiranza, Alnylam)
Major advances in sequencing, which has enabled the mapping of genetic drivers of diseases ranging from cancer to neurodegeneration
ASOs have been in development over the last 3 decades or so - will Molecular Biosystems getting the first patent in 1991 and the first trial initiated in 1992. However, 2016 was a major breakthrough for the field when Spiranza (from Ionis) was approved to treat spinal muscular atrophy. Now over 10 ASOs have been approved with a lot more in the pipeline. The two companies I admire doing work in the field are Arrowhead and Alnylam (which deserves a case study on aggregating IP early-on to win)
So how do ASOs compare with other major modalities:
Discovery
ASO - rapid; just need a target and a biological hypothesis. There is some medchem focused on 3 parts: nucleotides, sugar backbone, and phosphodiester bonds.
Small molecules - long time; screening then lead optimization
Biologics - long time as well; generate biologics, screen, optimize
MoA
ASO - single target; local delivery
Small molecules - single target most of the time where they inhibit a target through competitive binding; potential off-target effects and systemic delivery
Biologics - single target where they bind a target with high specificity
Clinical development
ASO - one NME with multiple actions; new modality so regulatory controls still being mapped out
Small molecules - NCE with one action; combinations easily available and regulatory path very clear
Biologics - NCE with one action; similar as small molecules
Manufacturing
ASO - standardized
Small molecules - synthetic chemistry; could be hard
Biologics - develop a cell line and process; very individualized for each drug
Synthesizing all this together, once an ASO and its backbone chemistry is designed, the sequence it targets is easily interchangeable. So the role of bioinformaticians and data scientists in ASOs is equivalent to the role medicinal chemists have in small molecule discovery. With this framing, an ASO’s pharmacokinetics becomes a lot more important than its ability to engage a target - https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5517098/ So what are large business opportunities in ASOs:
Adding new chemistries to ASO backbone to improve distribution and safety - https://www.idtdna.com/pages/education/decoded/article/an-aso-modification-that-enhances-nuclease-resistance-lowers-toxicity-and-increases-binding-affinity
Increasing stereopurity; ASOs can have many chiral centers generating (bases x linkages) number of diastereomers
Using software and data to predict distribution and toxicity for a given ASO sequence
Change FDA trial control requirements for ASOs and run more trials in parallel
Combining genomics-driven drug development with ASO development
Seven Powers cont.
The fifth of the seven powers is branding. What is a brand? For me, this is the hardest power to truly understand. A brand seems to have a long-term track record (i.e. reinforcing actions) around quality and experiences (i.e. evoke a positive emotion) that enables a business or product to be perceived as higher value than equivalents. But the long-term track record part can be taken from others through association as well. A company doesn’t necessarily need it initially to build a brand. An example is Apple associating itself with Gandhi and MLK early on. Brand provides benefits to a product mainly in two ways:
Effective valence - the feeling from the brand generates a positive feeling detached from the value of the product
Uncertainty reduction - customers will get what they expect
What is branding in life sciences? It’s often involved in consumer products particularly those with negative long-tail events. In drugs, branded mainly means patent protection not in the common use of the word. Generic drugs like aspirin are the major beneficiary of a brand because bad formulation or manufacturing contaminants drive people to continue to buy aspirin from Bayer and Viagra from Pfizer a bit.
Branding enables a company to have pricing power. However, companies growing this power have to be aware of brand dilution, geographic/cultural limits, changing preferences from their customers, and counterfeiting. Branding seems to be a very difficult power to use in life sciences. But as consumer products from biology (i.e. pet dragon) become more and more widespread, branding will become a lot more important in the industry.



Curious what do you mean by point 4 in the ASO portion. What would the would a change in FDA trial control requirements for ASOs and run more trials in parallel look like"
Personally, I think ASO cos should take a lesson from precision onc and run basket trials often to test different approaches and segment population.