More well thought out work can be found at — https://axial.substack.com/
Axial partners with great founders and inventors. We invest in early-stage life sciences companies often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialsprawl.com
New medicines #12 - July 25, 2020 - July 31, 2020
A weekly overview of the development of new medicines.
- Bluebird Bio with Bristol Myers Squibb announced the resubmission of their Biologics License Application (BLA) to the FDA for idecabtagene vicleucel (ide-cel; bb2121), for the treatment of adult patients with relapsed and refractory multiple myeloma - https://www.businesswire.com/news/home/20200729005776/en/Bristol-Myers-Squibb-bluebird-bio-Announce-Submission
- Genocea Biosciences released preliminary data from its Phase 1/2a trial evaluating its neoantigen vaccine GEN-009, combined with immune checkpoint inhibitor-based regimens in patients with solid tumors:
Three of five patients achieved separate RECIST responses after GEN-009 administration
- Onconova Therapeutics announced that the required number of survival events for the pivotal Phase 3 INSPIRE trial has been reached - https://www.globenewswire.com/news-release/2020/07/29/2069696/0/en/Onconova-Therapeutics-Announces-that-the-Required-Number-of-Survival-Events-Has-Been-Reached-for-the-Pivotal-Phase-3-INSPIRE-Trial-Data-Analysis.html
- Roche announced that its phase 3 trial of Actemra (tocilizumab; anti-IL-6) did not meet its primary endpoint of improved clinical status in hospitalized adult patients with severe COVID-19 associated pneumonia - https://www.businesswire.com/news/home/20200728006091/en/Genentech-Update-Phase-III-COVACTA-Trial-Actemra
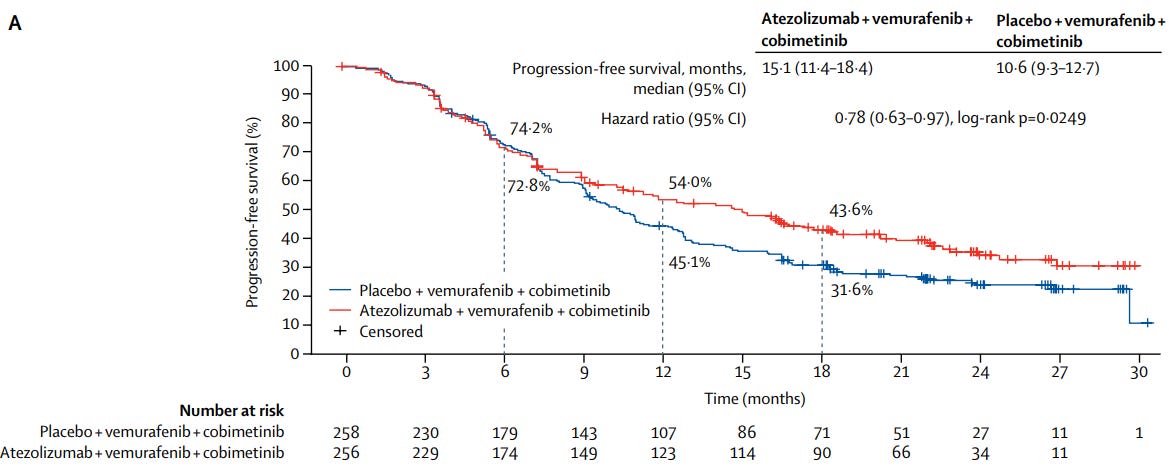
Moreover, Roche announced that the FDA approved Tecentriq (atezolizumab) plus Cotellic (cobimetinib) and Zelboraf (vemurafenib) for the treatment of BRAF V600 mutation-positive advanced melanoma patients:
Helped people live longer without disease worsening or death compared to placebo plus Cotellic and Zelboraf (median PFS 15.1 months versus 10.6 months respectively; hazard ratio, HR=0.78; confidence interval: 0.63-0.97; P=0.025)
The most common adverse reactions (rate ≥20%) in patients who received Tecentriq plus Cotellic and Zelboraf were rash (75%), musculoskeletal pain (62%), fatigue (51%), hepatotoxicity (50%), pyrexia (49%), nausea (30%), pruritus (26%), edema (26%), stomatitis (23%), hypothyroidism (22%), and photosensitivity reaction (21%)

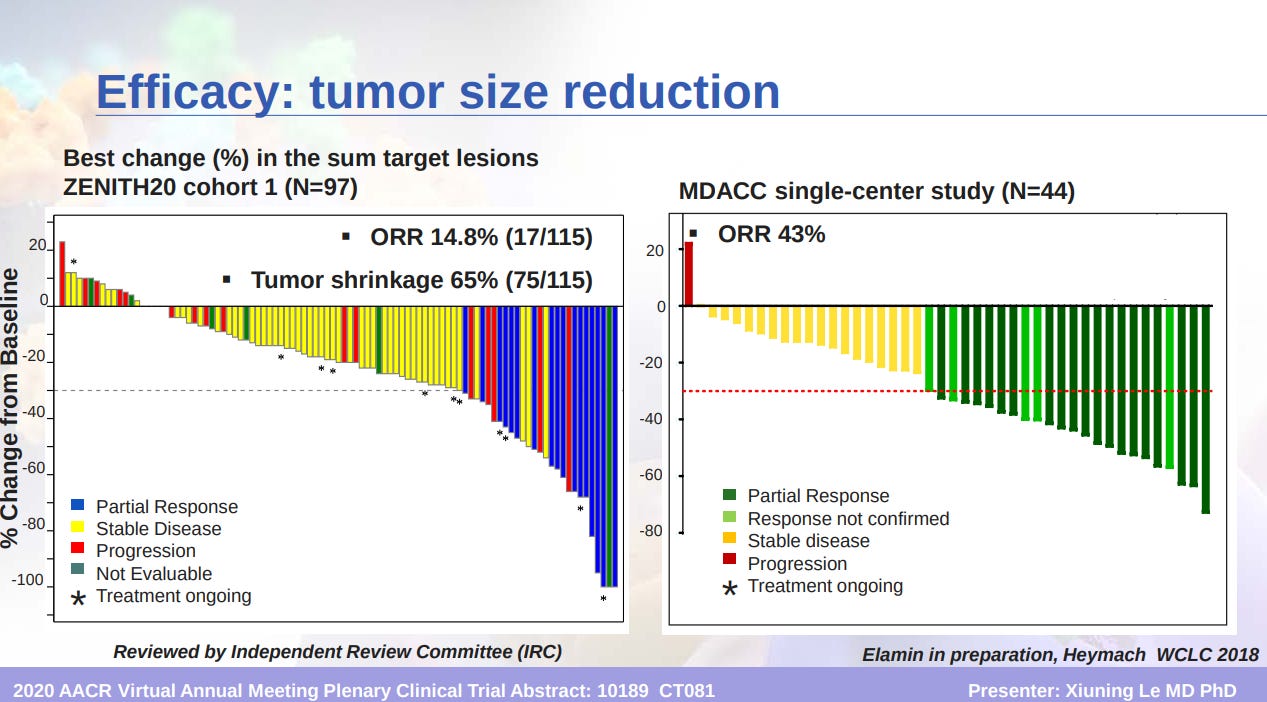
- Spectrum Pharmaceuticals announced that it met the pre-specified primary endpoint in the phase 2 clinical trial evaluating poziotinib in previously treated non-small cell lung cancer (NSCLC) patients with HER2 exon 20 insertion mutations (Cohort 2):
Objective response rate (ORR) of 27.8%

- TCR2 Therapeutics announced that patients showed unconfirmed partial responses in its phase 1 trial of TC-210 in patients with mesothelin-expressing solid tumors:
Two other patients exhibited stable disease through six months
https://finance.yahoo.com/news/tcr2-therapeutics-releases-positive-solid-143407440.html
Source: BPC, SY, Bioworld


